Entangled Molecules Capture Sulfate – Future solutions for water purification and environmental monitoring

Sulfate is notoriously difficult to bind in aqueous environments. It strongly prefers to remain surrounded by water molecules rather than attach to synthetic receptors, which is why only a handful of artificial systems can recognize sulfate effectively in water. Now researchers from the University of Jyväskylä have developed new receptors that overcome this challenge, binding sulfate more than a thousand times more strongly than most existing receptors in pure water.
- Performance of the receptors rivals that of natural protein binding sites, which are able to capture even extremely low concentrations of sulfate from their surroundings, rejoices Associate Professor Fabien Cougnon from the University of Jyväskylä.
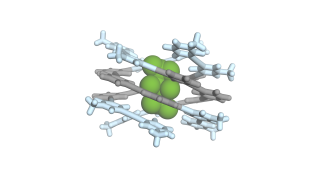
Interlocked “Solomon link” structures enable sulfate capture
This exceptional binding ability arises from the unusual architecture of the molecules. The research team designed structures known as “Solomon links”, molecules composed of two rings intricately interlocked and tied multiple times around each other. This mechanical entanglement creates a well-defined yet flexible cavity, in which positively charged regions and hydrogen-bonding groups work together to attract sulfate ions.
- Because the two rings are physically locked together, the binding cavity naturally adopts a shape that is well suited to sulfate. This structural “preorganization” means the molecule needs very little energy to adjust during binding, which is a key reason for its unusually high performance, explains Cougnon.
The study was published in the Journal Chem.
Article information:
- Multiply Entangled Receptors for High‑Affinity Anion Recognition in Water, Chem (Cell Press), 10.3.2026.
- Link to article: https://www.sciencedirect.com/science/article/pii/S2451929426000021
- DOI number: https://doi.org/10.1016/j.chempr.2026.102936