Mapping and modeling skeletal muscle properties in children and young adults with cerebral palsy (MaMo)
The MOVIE Lab aims to shift the focus toward understanding muscular adaptations in neuromuscular disease. We investigate how active and passive muscle–tendon properties interact to determine voluntary force generation, challenging traditional muscle models by proposing that losses in active force may be partially compensated by increased passive force contributions. By combining advanced 3D imaging and computational modeling, we seek to uncover fundamental compensation mechanisms in skeletal muscle, with cerebral palsy as the primary model condition and broader implications for other neurological disorders.
Project description
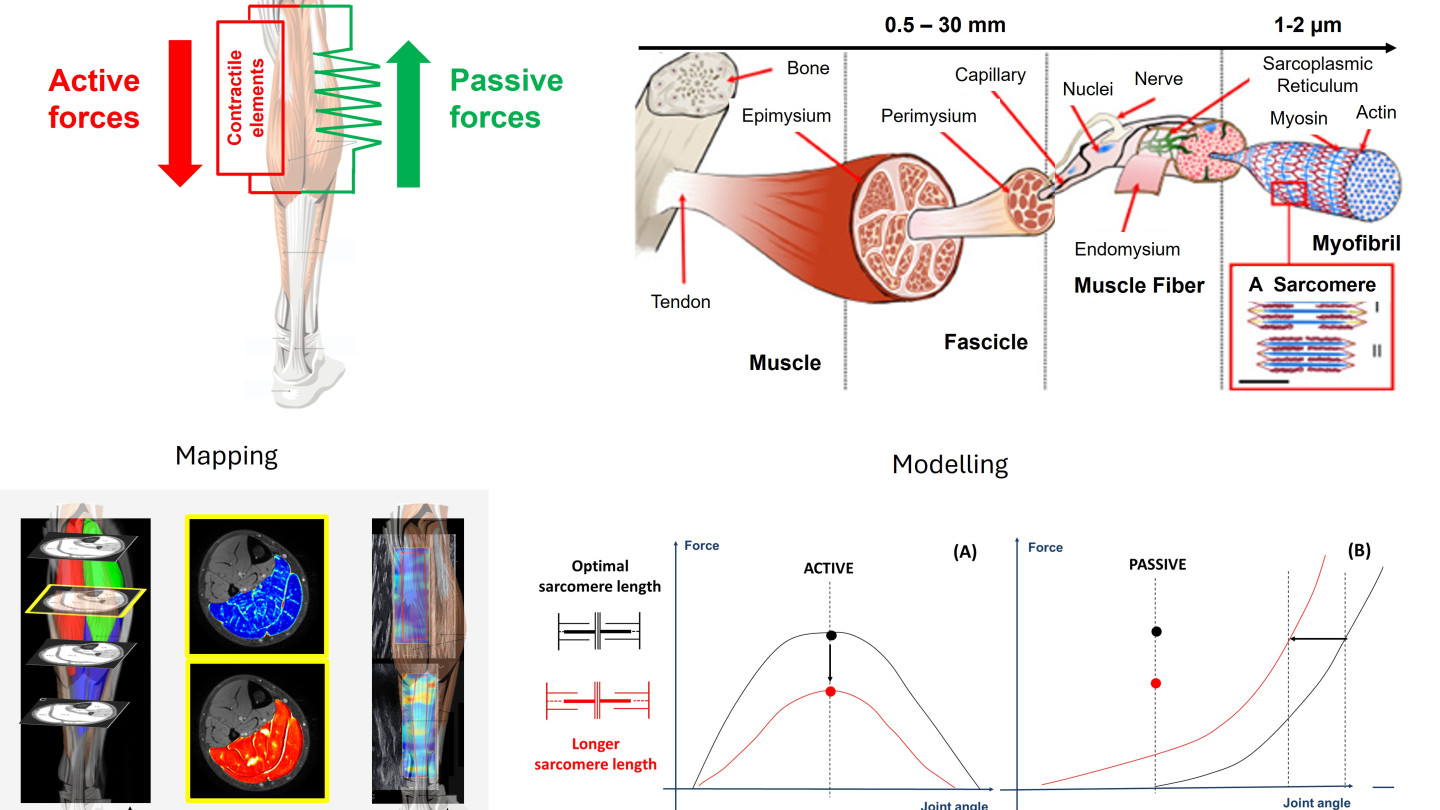
Human movement depends on the precise interaction between neural control and the mechanical properties of muscles and tendons. In neuromuscular disorders such as cerebral palsy (CP), reduced voluntary force production and functional limitations are traditionally attributed to impaired neural activation. However, emerging evidence suggests that intrinsic structural and mechanical adaptations within skeletal muscle may play a critical and previously underappreciated role.
We aim to investigate how active and passive muscle–tendon properties interact to regulate force generation during movement. Specifically, we will (1) map three-dimensional morphological and compositional muscle–tendon properties in cerebral palsy using advanced multimodal imaging, and (2) develop computational models to quantify compensatory mechanisms between active and passive force contributions. By integrating 3D ultrasonography, quantitative MRI, motion analysis, high-density electromyography, and personalized muscle–tendon modeling, we seek to uncover how structural remodeling influences functional output across biological scales.
For the first time, we propose to challenge the traditional Hill-type muscle model by introducing a new paradigm in which loss of active force may be partially compensated by increased passive force transmission. This project will provide fundamental insights into muscular adaptation mechanisms in cerebral palsy and establish a framework transferable to other neurological and musculoskeletal conditions.
Imaging and motion analysis measurements are conducted using the Motion Analysis Laboratory at the Faculty of Sport and Exercise Sciences and MRI facilities at Hospital Nova. Clinical data collection is performed in close collaboration with the New Children’s Hospital in Helsinki. International collaborations include KU Leuven (Prof. Kaat Desloovere) and KTH Royal Institute of Technology (Prof. Lanie Gutierrez and Prof. Ruoli Wang).